Coenzyme A Elisa
Accession Number : KLM0000090 This work is released into the public domain; please see our release statement.
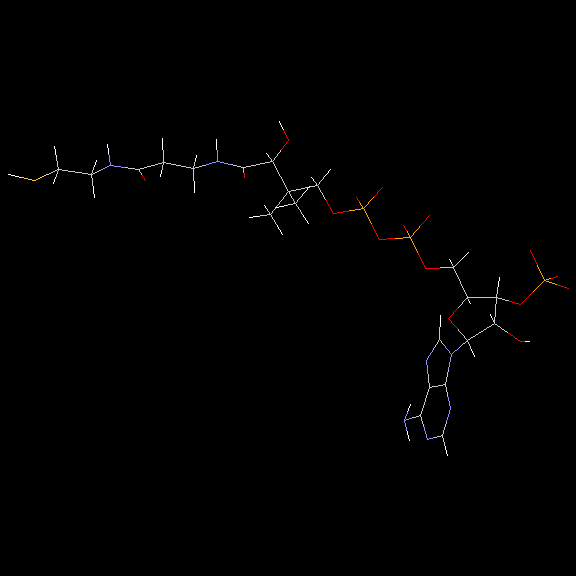
Doug Markham has contributed a molecular mechanics computation of the structure! See below for the details.
Coenzyme A (CoA) is an acyl group transfer coenzyme involved in numerous metabolic pathways such as the Krebs cycle and beta-oxidation. It was first isolated in 1951 by the German biochemist Feodor Lynen (who was awarded the Nobel Prize in 1964) in the form of acetyl-coenzyme A ("activated acetic acid") from yeast cells.
Synonyms :
CoA
CoASH
COENZYME-A
COA
Config Rule :
% coenzymeA
config(coenzymeA,[
substituent('R-pantothenyl'),
substituent('3''-phospho-ADP-beta-yl'),
substituent('beta-mercaptoethylamino-N-yl'),
linkage(from('beta-mercaptoethylamino-N-yl',nit(1)),
to('R-pantothenyl',car(1)),
trans(car(2),car(2)),cn_resonant),
linkage(from('R-pantothenyl',oxy(4)),
to('3''-phospho-ADP-beta-yl',pho(3)),
right,single)]).
config('3''-phospho-ADP-beta-yl',[
substituent('D-1-dehydroxy-3-phospho-5-oxy-ribofuranosyl'),
substituent(adenyl),
substituent(diphosphopentaoxygen),
linkage(from('D-1-dehydroxy-3-phospho-5-oxy-ribofuranosyl',car(1)),
to(adenyl,nit(9)),
up,single),
linkage(from('D-1-dehydroxy-3-phospho-5-oxy-ribofuranosyl',attach_to([oxy,car(5)])),
to(diphosphopentaoxygen,pho(1)),
nil,single)]).
config('D-1-dehydroxy-3-phospho-5-oxy-ribofuranosyl',[
ring([
oxy,
anomeric(1,hyd),
car(2,hyd&&hydroxyl;),
car(3,hyd&&phosphate;),
car(4,oxymethyl&&hyd;)])]).
config(adenyl,[
model(adenine,[
diff(nit(9,hyd),nit(9))])]).
config(adenine,[
model(purine,[
diff(car(6,hyd),car(6,amine(10)))])]).
config(purine,[
ring_system([
ring([
car(6,hyd)&,
car(5)&,
car(4)&,
nit(3)&,
car(2,hyd)&,
nit(1)&]),
ring([
nit(7)&,
car(8,hyd)&,
nit(9,hyd)&,
car(4)&,
car(5)&])],
conjugate(1,pseudopos([car(4),car(5)]),2,pseudopos([car(4),car(5)]))])]).
config('R-pantothenyl',[
chain([
car(1,oxy?),
car(2,hyd&&hyd;),
car(3,hyd&&hyd;),
nit(1,hyd)#,
car(4,oxy?),
car(5,hydroxyl&&hyd;),
car(6,methyl&&methyl;),
oxymethyl]),
trans(car(3),car(5),bond(nit(1),car(4)))]).
config('beta-mercaptoethylamino-N-yl',[
chain([
sul(1,hyd),
car(1,hyd&&hyd;),
car(2,hyd&&hyd;),
nit(1,hyd)])]).
Smiles String :
[C@2H2]([C@2H2]/[NH]=[C](/[C@2H2][C@2H2]/[NH]=[C](/[C@2H]([OH])[C@2]([CH3])([CH_
3])[C@2H2][O][P@2]([O][P@2](=[O])([O][C@2H2][C@1H]1([C@2H]([O][P@2](=[O])([O-])_
[O-])[C@2H]([OH])[C@2H]([O]1)-[n]1([cH][n][c]2([c]1[n][cH][n][c]2[NH2]))))[O-])_
([O-])=[O])\[O])\[O])[SH]
coenzymeA
Terminal :
% coenzymeA
c(1,12,(0,nonchiral))-[c(2,left)~,h(2,right)~,s(1,up)~,h(3,down)~],
c(2,12,(0,nonchiral))-[h(4,left)~,c(1,right)~,h(5,up)~,n(1,(down,isomeric(up)))~],
c(3,12,(0,nonchiral))-[c(4,(right,isomeric(up)))~,o(2,(nil,isomeric(down)))?,n(1,trans)#], c(4,12,(0,nonchiral))-[c(3,(left,isomeric(down)))~,c(5,right)~,h(8,up)~,h(7,down)~],
c(5,12,(0,nonchiral))-[c(4,left)~,h(9,right)~,h(10,up)~,n(2,(down,isomeric(up)))~],
c(6,12,(0,nonchiral))-[o(3,(nil,isomeric(down)))?,c(7,(down,isomeric(up)))~,n(2,trans)#], c(7,12,(0,chiral))-[o(1,left)~,h(12,right)~,c(6,(up,isomeric(down)))~,c(8,down)~],
c(8,12,(0,nonchiral))-[c(9,left)~,c(11,right)~,c(7,up)~,c(10,down)~],
c(9,12,(0,nonchiral))-[h(14,left)~,c(8,right)~,h(15,up)~,h(16,down)~],
c(10,12,(0,nonchiral))-[h(17,left)~,h(19,right)~,c(8,up)~,h(18,down)~],
c(11,12,(0,nonchiral))-[c(8,left)~,h(20,right)~,o(4,up)~,h(21,down)~],
c(12,12,(0,chiral))-[c(13,left)~,o(5,right)~,n(11,up)~,h(22,down)~],
c(13,12,(0,chiral))-[c(14,left)~,c(12,right)~,h(23,up)~,o(6,down)~],
c(14,12,(0,chiral))-[c(15,left)~,c(13,right)~,h(25,up)~,o(7,down)~],
c(15,12,(0,chiral))-[o(5,left)~,c(14,right)~,c(16,up)~,h(26,down)~],
c(16,12,(0,nonchiral))-[o(11,left)~,h(27,right)~,h(28,up)~,c(15,down)~],
c(17,12,(0,nonchiral))-[h(31,nil)~,n(5,flat)&,n(3,flat)&],
c(19,12,(0,nonchiral))-[n(5,flat)&,c(20,flat)&,n(11,flat)&],
c(20,12,(0,nonchiral))-[c(19,flat)&,c(21,flat)&,n(9,flat)&],
c(21,12,(0,nonchiral))-[n(12,nil)~,n(3,flat)&,c(20,flat)&],
c(23,12,(0,nonchiral))-[h(32,nil)~,n(9,flat)&,n(11,flat)&],
h(1,1,(0,nonchiral))-[s(1,nil)~],
h(2,1,(0,nonchiral))-[c(1,left)~],
h(3,1,(0,nonchiral))-[c(1,up)~],
h(4,1,(0,nonchiral))-[c(2,right)~],
h(5,1,(0,nonchiral))-[c(2,down)~],
h(6,1,(0,nonchiral))-[n(1,(nil,isomeric(down)))~],
h(7,1,(0,nonchiral))-[c(4,up)~],
h(8,1,(0,nonchiral))-[c(4,down)~],
h(9,1,(0,nonchiral))-[c(5,left)~],
h(10,1,(0,nonchiral))-[c(5,down)~],
h(11,1,(0,nonchiral))-[n(2,(nil,isomeric(down)))~],
h(12,1,(0,nonchiral))-[c(7,left)~],
h(13,1,(0,nonchiral))-[o(1,nil)~],
h(14,1,(0,nonchiral))-[c(9,right)~],
h(15,1,(0,nonchiral))-[c(9,down)~],
h(16,1,(0,nonchiral))-[c(9,up)~],
h(17,1,(0,nonchiral))-[c(10,right)~],
h(18,1,(0,nonchiral))-[c(10,up)~],
h(19,1,(0,nonchiral))-[c(10,left)~],
h(20,1,(0,nonchiral))-[c(11,left)~],
h(21,1,(0,nonchiral))-[c(11,up)~],
h(22,1,(0,nonchiral))-[c(12,up)~],
h(23,1,(0,nonchiral))-[c(13,down)~],
h(24,1,(0,nonchiral))-[o(6,nil)~],
h(25,1,(0,nonchiral))-[c(14,down)~],
h(26,1,(0,nonchiral))-[c(15,up)~],
h(27,1,(0,nonchiral))-[c(16,left)~],
h(28,1,(0,nonchiral))-[c(16,down)~],
h(29,1,(0,nonchiral))-[n(12,nil)~],
h(30,1,(0,nonchiral))-[n(12,nil)~],
h(31,1,(0,nonchiral))-[c(17,nil)~],
h(32,1,(0,nonchiral))-[c(23,nil)~],
n(1,14,(0,nonchiral))-[h(6,(nil,isomeric(up)))~,c(2,(up,isomeric(down)))~,c(3,trans)#], n(2,14,(0,nonchiral))-[h(11,(nil,isomeric(up)))~,c(5,(up,isomeric(down)))~,c(6,trans)#], n(3,14,(0,nonchiral))-[c(17,flat)&,c(21,flat)&],
n(5,14,(0,nonchiral))-[c(17,flat)&,c(19,flat)&],
n(9,14,(0,nonchiral))-[c(20,flat)&,c(23,flat)&],
n(11,14,(0,nonchiral))-[c(12,down)~,c(23,flat)&,c(19,flat)&],
n(12,14,(0,nonchiral))-[h(29,nil)~,h(30,nil)~,c(21,nil)~],
o(1,16,(0,nonchiral))-[h(13,nil)~,c(7,right)~],
o(2,16,(0,nonchiral))-[c(3,(nil,isomeric(up)))?],
o(3,16,(0,nonchiral))-[c(6,(nil,isomeric(up)))?],
o(4,16,(0,nonchiral))-[p(3,right)~,c(11,down)~],
o(5,16,(0,nonchiral))-[c(12,left)~,c(15,right)~],
o(6,16,(0,nonchiral))-[h(24,nil)~,c(13,up)~],
o(7,16,(0,nonchiral))-[p(1,right)~,c(14,up)~],
o(8,16,(-6.666666666666666E-01,nonchiral))-[p(1,left)?],
o(9,16,(-6.666666666666666E-01,nonchiral))-[p(1,up)?],
o(10,16,(-6.666666666666666E-01,nonchiral))-[p(1,down)?],
o(11,16,(0,nonchiral))-[c(16,right)~,p(2,up)~],
o(12,16,(0,nonchiral))-[p(3,left)~,p(2,right)~],
o(13,16,(-5.0E-01,nonchiral))-[p(2,left)?],
o(14,16,(-5.0E-01,nonchiral))-[p(2,down)?],
o(15,16,(-5.0E-01,nonchiral))-[p(3,up)?],
o(16,16,(-5.0E-01,nonchiral))-[p(3,down)?],
p(1,31,(0,nonchiral))-[o(7,left)~,o(8,right)?,o(10,up)?,o(9,down)?],
p(2,31,(0,nonchiral))-[o(12,left)~,o(13,right)?,o(14,up)?,o(11,down)~],
p(3,31,(0,nonchiral))-[o(4,left)~,o(12,right)~,o(16,up)?,o(15,down)?],
s(1,32,(0,nonchiral))-[h(1,nil)~,c(1,down)~]
The Terminals for all the Config Rules are in Prolog Definite Clause Grammar (DCG) form.They can be checked in the Manual here.
The compound's PDB file can be seen here.
Doug Markham of the Institute for Cancer Research, Fox Chase Cancer Center,Philadelphia, PA, has contributed the following structure for coenzymeA. He computed this structure in sdf format using MacroModel, a molecular mechanics program. We have used Babel to convert the .sdf format to PDB format. You'll find it interesting to compare these structures to those computed using CONCORD.
Many thanks Doug!
